Artificial sweeteners are confusing. They have weird names and are metabolized (broken down) by the body into components whose names we’re not familiar with. That makes them scary. So, in anticipation of an upcoming post about the War on Artificial Sweeteners, let’s review the current artificial sweeteners on the market today.

The amount of sugar (on the left) versus aspartame (on the right) that it would take to sweeten 2 quarts of Kool-Aid.
Disclaimer: I am not suggesting that you increase your consumption of artificial sweeteners. Nor am I advocating or endorsing one sweetener over another. My aim here is simply to provide scientific answers to some of the common concerns I often encounter. These are facts; not my opinions (although I understand my unintentional bias is likely found). You will find the sources I cited with links throughout.
Aspartame
Let’s just get this out of the way. Aspartame is probably the most feared of all the artificial sweeteners out there. Its being pulled from products left and right. Why? To be honest, I’m not really sure. There are many, many myths about aspartame that have all been refuted in hundreds of very robust research studies. It’s one of the most highly studied artificial sweeteners.
Before I tell you about aspartame, I must tell you about proteins. Proteins are made of building blocks called amino acids. There are 20 amino acids that are strung together in different orders and lengths that make up all the proteins in the world.
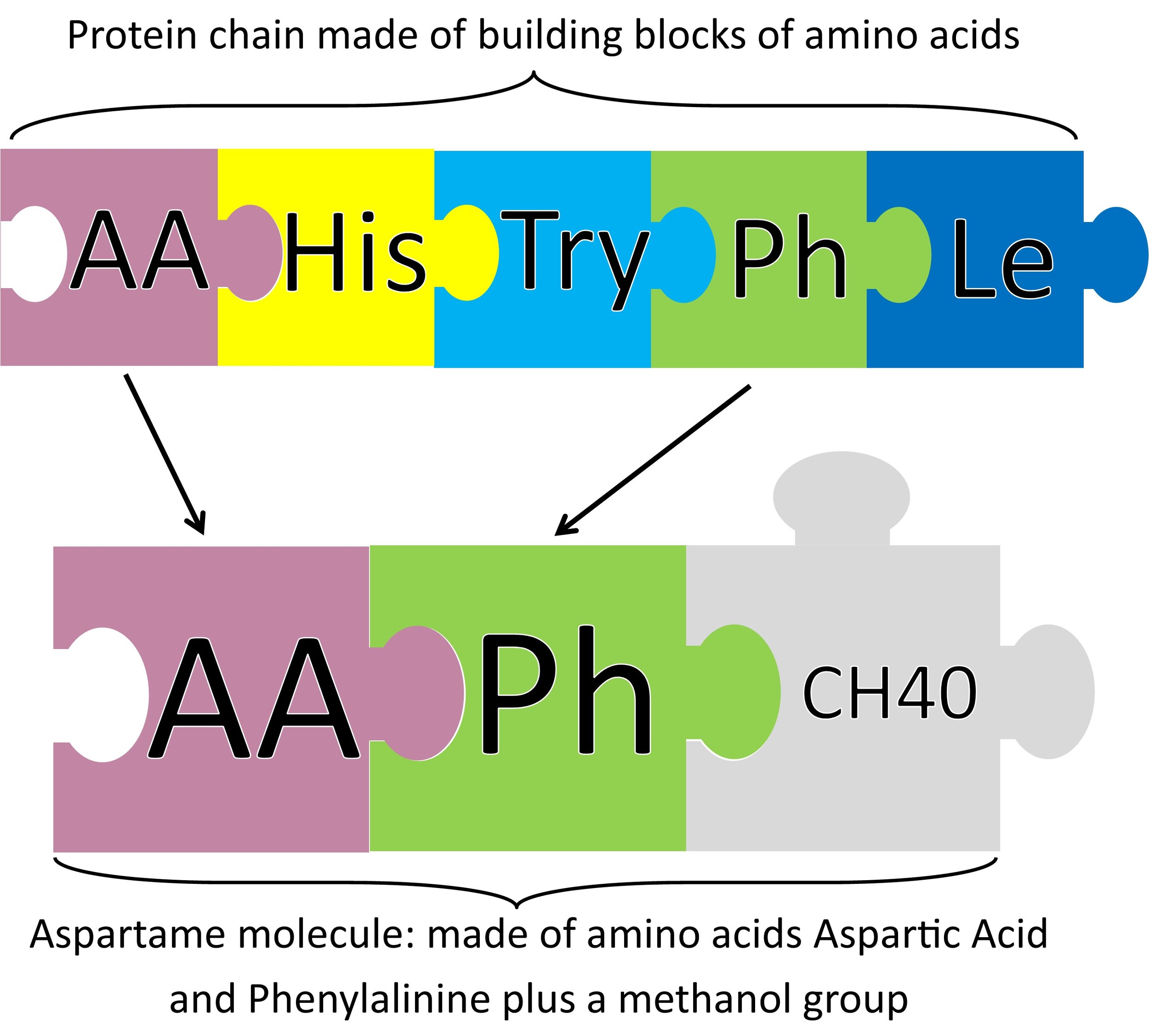
Aspartame is made of two of these amino acids: phenylalinine and aspartic acid, with a methanol group attached to the end. When metabolized it’s broken into its individual amino acids (just like protein), plus a little tiny bit of methanol (see myth below).
Aspartame is 200 times sweeter than sugar.
1 cup sugar = 1/4 teaspoon aspartame
The Acceptable Daily Intake (ADI) of aspartame is 50g/kg body weight. So, a 150 lb person could safely consume 81 packets of Equal per day.
Common Myths about Aspartame
Aspartame breaks down into methanol (wood alcohol) in your body. Okay, well this is actually true – methanol is a metabolite of aspartame. However, methanol is released in much higher amounts from eating fruits and vegetables. Methanol is only a problem in large amounts (say, for instance, poorly-made moonshine) – remember: the dose determines the poison. We eat really tiny amounts of aspartame, and so the amount of methanol that is produced is virtually nothing. In fact, studies have shown that eating aspartame does not increase blood levels of methanol. To put this in perspective, one liter of diet soda has about 55 milligrams of methanol, compared to about 680 milligrams of methanol in a liter of fruit juice. So, in reality, this is a non-issue.
Aspartame breaks down into formaldehyde in the body. Also technically true, but also not a concern. Formaldehyde is actually produced by our bodies in quantities thousands times greater than what you would consume from aspartame. Formaldehyde is actually also a necessary component of creating other important compounds in the body, including DNA. Long story short, this concern is not something to lose sleep over if you have aspartame in your diet.
Aspartame causes a bunch of bad stuff, ranging from headaches to cancer to seizures to nausea. There have been many, many studies about this and none have shown any evidence of this to be true. Quality studies. Double-blind, placebo-controlled studies. It would take many paragraphs to review all of the literature about this, so if you want more info on the studies, take a look at this overview. More likely what causes “symptoms” from consuming aspartame is a placebo effect.
People with phenylketonuria (PKU) can’t have aspartame because it breaks down into phenylalinine which builds up to dangerous blood levels. Actually, this is also true. PKU is a genetic disorder in which a person can’t metabolize the amino acid phenylalinine. However, phenylaline is a very common amino acid found in most normal protein-containing foods. A person with PKU has to avoid way, way more than just aspartame and eats a very specific diet.
Neotame
Neotame is pretty new. It’s not in many products (although I found it in my favorite lite yogurt), but I think we’ll soon be finding it in all sorts of things.
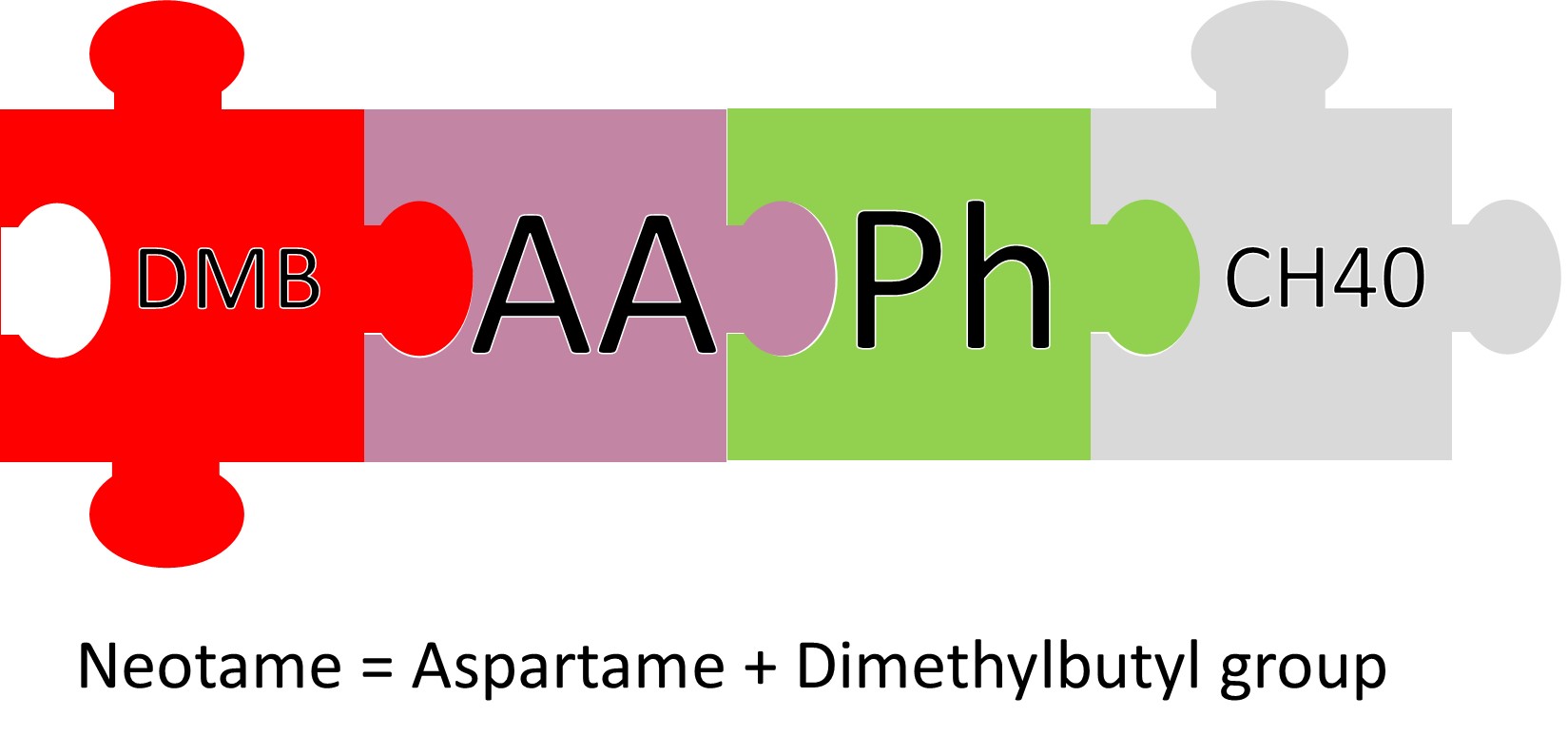
Neotame is a derivative of aspartame. It’s actually a molecule of aspartame with a dimethylbutanyl (DMB) group attached. I know that sounds crazy, but that little DMB enhances the sweetness of aspartame 30-fold.
Neotame is 7,000 – 13,000 times sweeter than sugar.
1 cup sugar is less than 1/100th teaspoon neotame
The Acceptable Daily Intake (ADI) of neotame is 0.3g/kg body weight. So, a 150 lb person could safely consume 23 packets of sweetener per day.
Common Myths about Neotame
Myth: It’s way worse than aspartame. No, it’s not. In fact, it’s probably safer than aspartame because you need way less of it. Remember that thing about the dose determines the poison? Your body breaks down neotame and it’s excreted in the urine and feces. So no worries – the tiny, tiny amount of neotame you consume is all taken care of.
Advantame
Approved by the FDA in 2014, advantame is one of the newest sweeteners available on the market. In fact, it’s not in many foods yet – less than neotame.
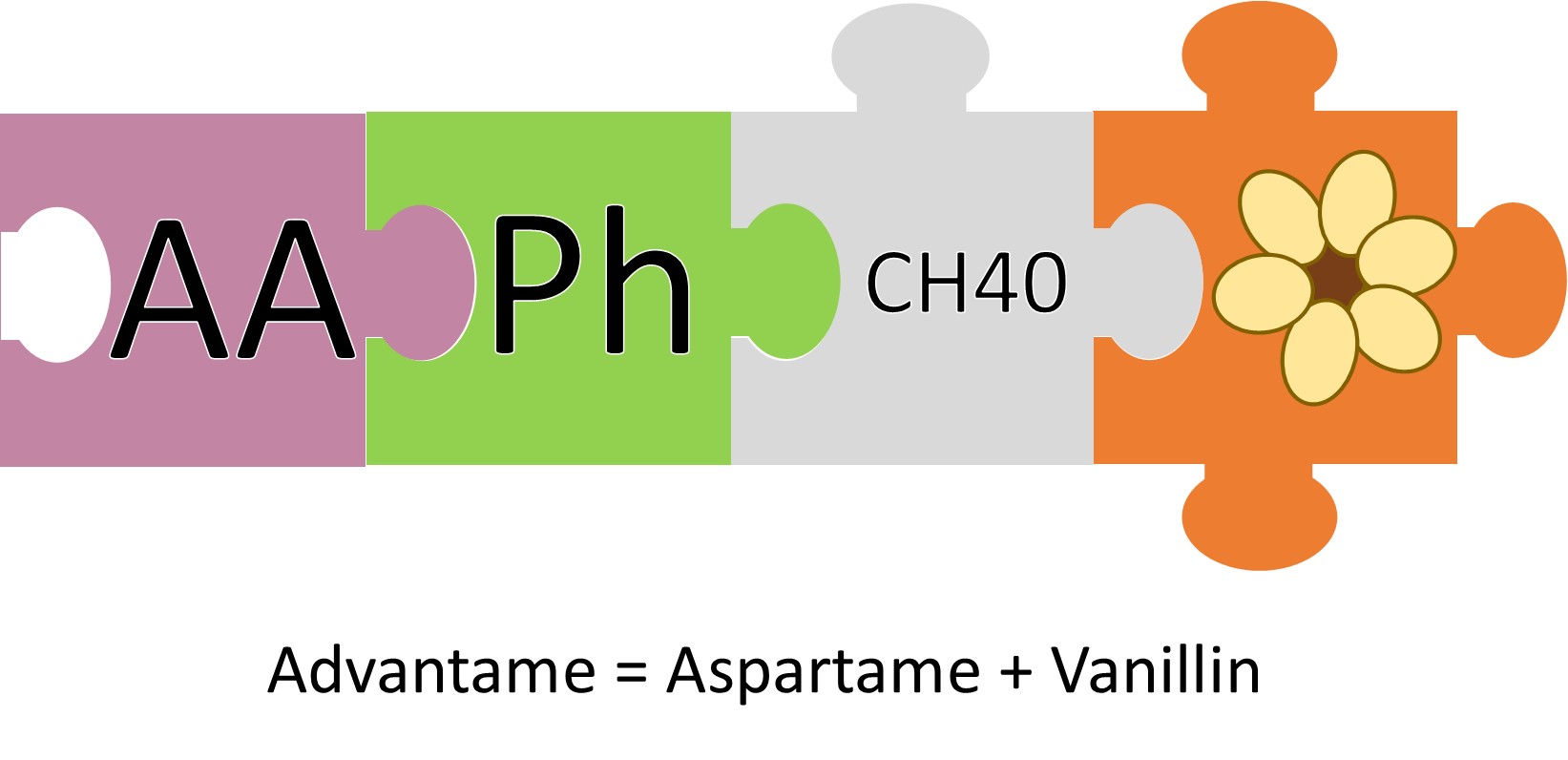
Much like neotame, advantame is a derivative of aspartame. It’s an aspartame molecule with vanillin (yes, the molecule that makes vanilla taste like vanilla). Somehow, this vanillin makes the aspartame crazy sweet. One hundred times sweeter than aspartame, in fact.
Advantame is 20,000 times sweeter than sugar.
1 cup sugar is less than 1/200th teaspoon advantame
The Acceptable Daily Intake (ADI) of advantame is 32.8g/kg body weight. So, a 150 lb person could safely consume 4,920 packets of sweetener per day.
Common Myths about Advantame
Myth: It’s way worse than aspartame. No, it’s not. Similar to neotame, you need so very little advantame that it’s no trouble for your body to metabolize it without harm. Advantame is poorly absorbed by the body, so it’s mostly excreted in the feces.
Sucralose
Found in the lovely little yellow packets, it seems to me that sucralose has become the darling of the artificial sweetener world lately – probably as a result of the fears about aspartame.
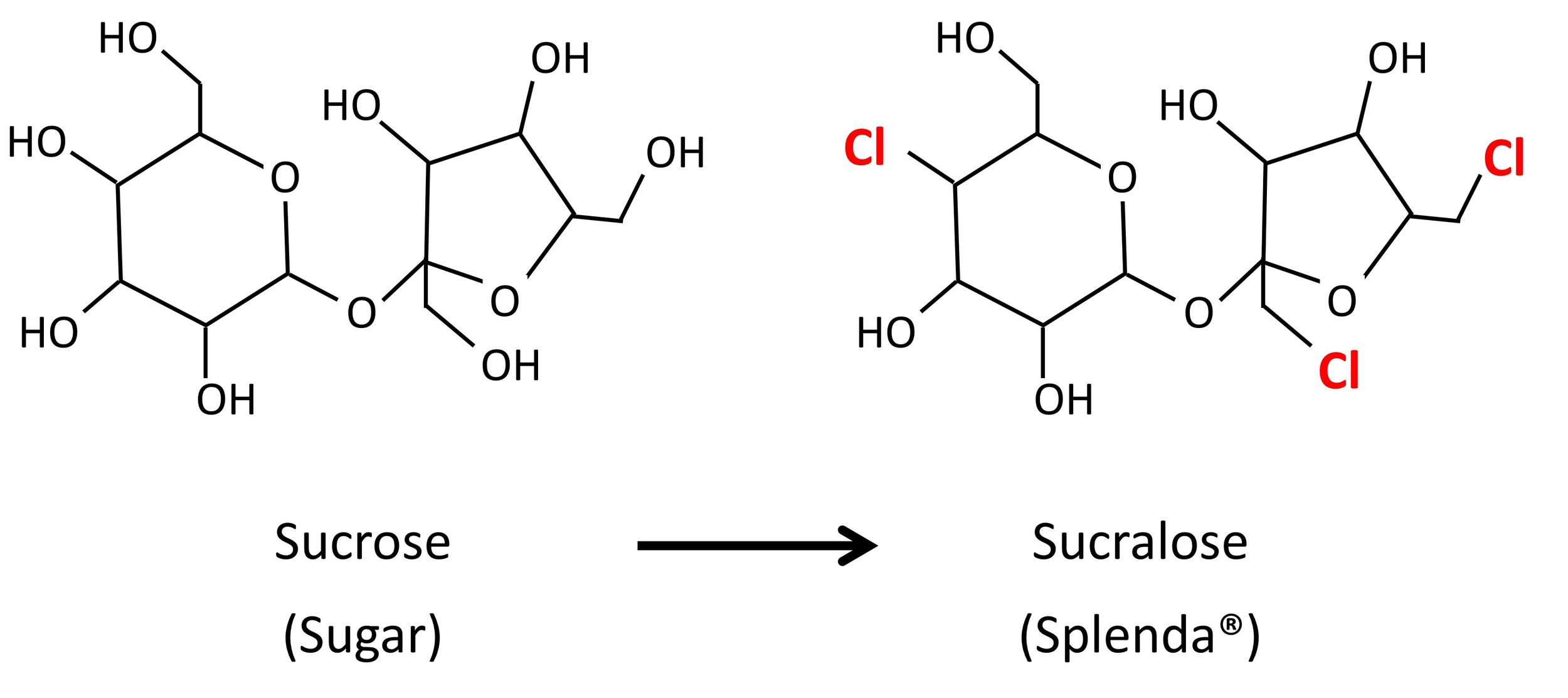
Sucralose is a sugar (sucrose) molecule that has three of the -OH (hydroxyl) groups switched out for chloride molecules. This has the effect of making the molecule super sweet.
Sucralose is 600 times sweeter than sugar.
1 cup sugar = 1/10th teaspoon sucralose
The Acceptable Daily Intake (ADI) of sucralose is 5g/kg body weight. So, a 150 lb person could safely consume 28 packets of Splenda per day.
Common Myths about Sucralose
Sucralose breaks down into chlorine, which is dangerous. No, that’s not true. Sucralose is poorly absorbed by the body (only about 15% is actually absorbed, thus why it is essentially calorie-free) and no chlorine molecules are liberated. Also, just because something is dangerous in one compound doesn’t mean it’s dangerous in all circumstances. Yes, chlorine gas is dangerous. However, chlorine as part of sodium chloride (table salt) is not. In moderation, sucralose is perfectly safe, a statement that is supported by 110 studies that were submitted to the FDA for approval.
Sucralose alters the gut bacteria and may lead to obesity. There is no evidence of this. The study published in The Journal of Toxicology turned out to be funded by the Sugar Association (conflict of interest, anyone?) and a year later was refuted by an expert panel that determined the claims were not substantiated by current published literature or the data provided.
Saccharin
Saccharin is the oldest artificial sweetener on the market today. When sugar shortages became widespread and the price spiked, saccharin became commonly used in both world wars as a sweetener.
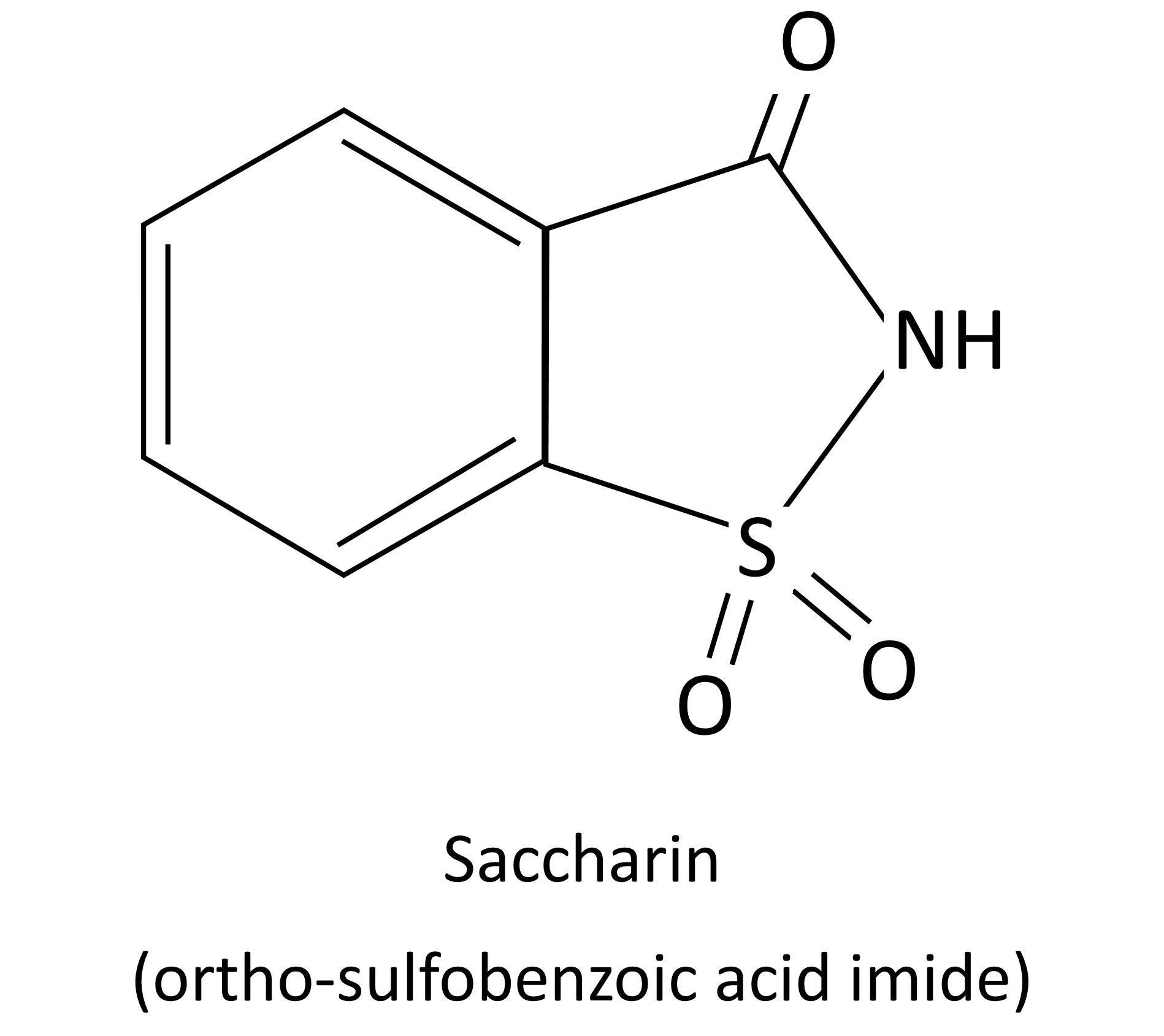
According to the Encyclopedia Britannica, saccharin is “An organic compound employed as a non-nutritive sweetening agent. It occurs as insoluble saccharin or in the form of various salts, primarily sodium and calcium.” Its technical name is ortho-sulfobenzoic acid imide.
It was randomly discovered in a lab at John Hopkins University when a researcher (accidentally?) licked his finger and found the compound he just made was sweet. For the record, if you ever create an unknown compound, you should probably wash your hands rather than lick them. Those guys got lucky.
Saccharin is 300-500 times sweeter than sugar.
1 cup sugar = 1/8th teaspoon saccharin
The Acceptable Daily Intake (ADI) of saccharin is 15g/kg body weight. So, a 150 lb person could safely consume 28 packets of Sweet n’Low per day.
Common Myths about Saccharin
Myth: Saccharin causes bladder cancer. In rats. In the 1970s, a study found that giving large amounts (1% of the diet) of saccharin to rats caused them to develop bladder cancer. However, it was later found that the mechanism by which rats develop this type of cancer is different than in humans, so the results are really not applicable in us homo sapiens.
Acesulfame Potassium
Also known as Acesulfame-K or Ace-K, this is a super-sweet chemical compound. It’s absorbed from the digestive system but not metabolized and is excreted in the urine. Thus, we don’t get any calories from eating it.
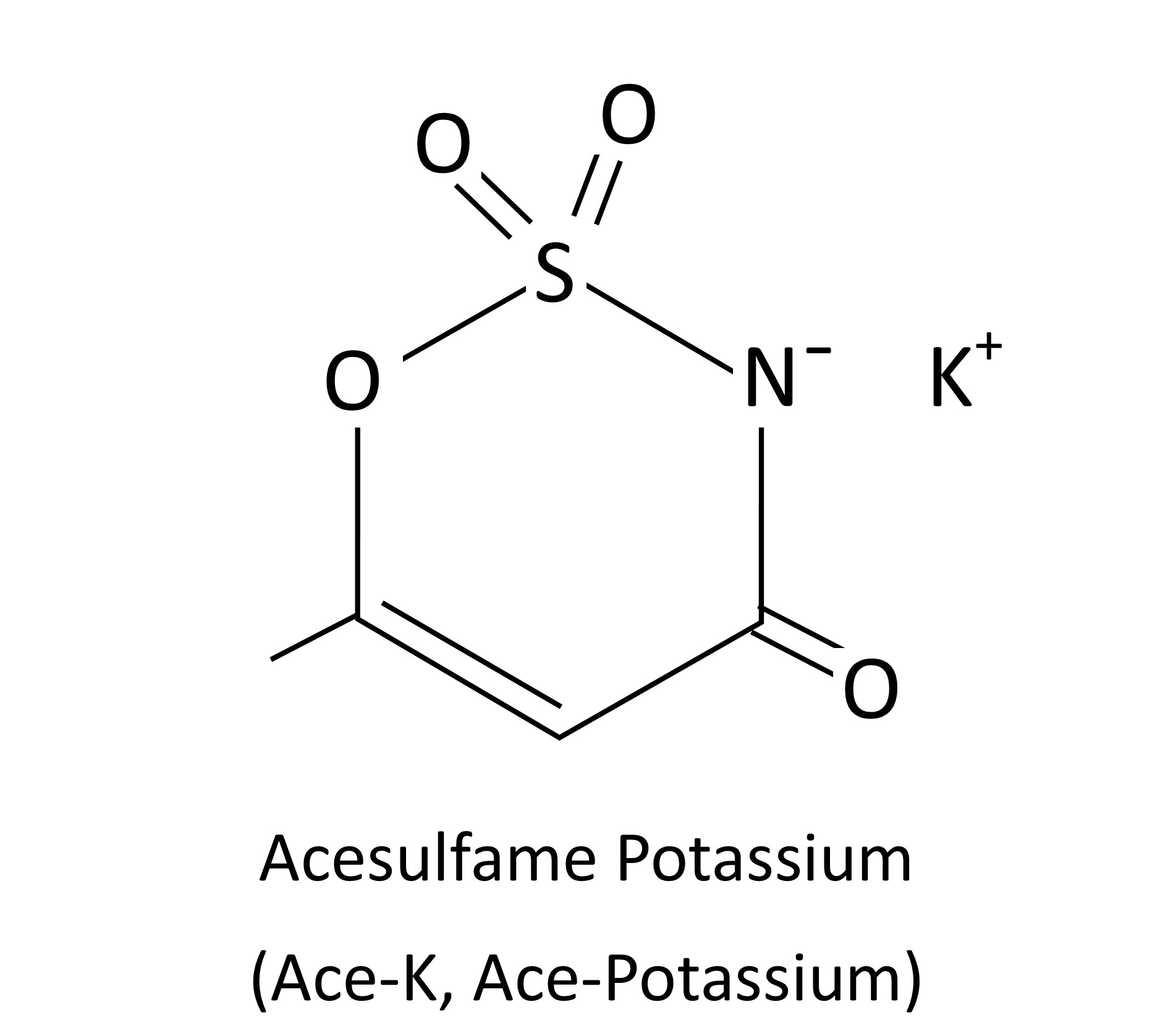
Ace-K has a clean, sweet taste with little aftertaste and tends to enhance the sweetness of other artificial sweeteners. You’ll often find it combined with other sweeteners.
Ace-K is 200 times sweeter than sugar.
1 cup sugar = 1/4th teaspoon ace-K
The Acceptable Daily Intake (ADI) of Ace-K is 15g/kg body weight. So, a 150 lb person could safely consume 26 packets of Sweet Ones per day.
Common Myths about Acesulfame Potassium
Acesulfame potassium contains methylene chloride, which is a carcinogen. Well, hardly. Methylene chloride is used as a solvent in the initial creation of Ace-K, but once purified the levels are so low it is no longer detectable. Also, remember how the dose determines the poison? The FDA has determined that up to 30 parts per billion of methylene chloride is safe, so even if there is a tiny amount, your body can handle it. Generally speaking, there have been many studies about Ace-K’s risk of cancer and they have all determined that there is no mutagenic or carcinogenic potential with the substance.
Stevia

Photo from http://sweetgreenfields.com/our-story/about-stevia/
A popular new sweetener because it is “natural,” stevia is derived from the stevia rebaudiana Bertoni plant. The plant is native to South America where its leaves have been used (as with Asia) as a sweetener for many years. It’s metabolized and excreted in the urine, so no calories for you!
The FDA approved stevioside as Generally Recognized as Safe (GRAS) in 2008 – meaning that experts in the field have reviewed the research and determined its safety, but the FDA itself has not reviewed the data. Stevioside is the pure form of the stevia plant; the FDA has not approved the use of the whole leaf as a sweetener due to concerns about the effect on various organ systems.
Stevia is 200-400 times sweeter than sugar.
1 cup sugar = 1/5th teaspoon stevia
The Acceptable Daily Intake (ADI) of stevia is 12g/kg body weight. So, a 150 lb person could safely consume 40 packets of Truvia per day.
Common Myths about Stevia
Stevia may cause all kinds of bad things. As I mentioned, the FDA has some concern about whole leaves from the stevia plant affecting different organ systems, including renal, reproductive, and cardiovascular. However, there has been much independent research on the purified forms of stevia, including Rebaudioside A (Reb A), Stevioside, Rebaudioside D, Steviol glycoside. These have been determined GRAS (Generally Recognized as Safe).
Monk Fruit (Luo Han Guo)

Photo from http://agedefensewater.com/about05.html
Monk fruit is the newest sugar substitute on the market today, as the FDA determined it to be GRAS in 2014. Monk fruit is a small melon native to Southern China. According to legend, the fruit was named after the Buddhist monks that cultivated it 800 years ago.
The juice of monk fruit is twenty times sweeter than that of other fruit. Unlike other fruit, the melon’s sweetness comes from an antioxidant it contains, mogroside V.
Monk fruit extract is 100-250 times sweeter than sugar.
1 cup sugar = 1/4th teaspoon monk fruit extract
The FDA hasn’t set an ADI for monk fruit extract, likely because the safe level of intake is way, way higher than what a person would consume in a day.
Common Myths about Monk Fruit
It’s not completely natural. I mean, I guess that’s true – most sweetener packets are packaged with an anti-caking agent, like dextrose, so it’s not pure monk fruit extract. Also, monk fruit must go through a purification process before becoming the extract used to sweeten beverages and foods. That same argument could be applied to cane sugar (even “raw” cane sugar), as it also goes through extensive processing for the sugarcane to be transformed into the sugar we buy in the store.
Additional Information
Want to learn more? Here are some additional (credible) references to learn more about artificial sweeteners:
The Food and Drug Administration
Hope this helps make more sense out of all of your sweet options. With love, from Peas and Hoppiness.
4 thoughts on “An Overview of Artificial Sweeteners”
Great information! I love reading your posts! I have heard that artificial sweeteners can increase appetite because your body releases insulin since it "thinks" that sugar is coming and therefore you crave sugary foods after consuming an artificial sweetener. Is this true at all? Thanks for all the good information! 🙂
Christine, you ask a great question! I will attempt to answer in a concise manner, as there is a plethora of research available about this topic. Let me separate your question into three parts.
1: Do artificial sweeteners increase your appetite? No. This has been most extensively studied with aspartame. Double-blind, placebo-controlled studies show that including artificial sweeteners in the diet in place of sugar either has a neutral or loss effect on weight. Recently, there have been some epidemiologic (population-based instead of individual) studies that have shown a correlation between higher intake of artificial sweeteners and increased body weight. However, there are so many variables that could contribute to this association (do people eat other unhealthy foods when drinking diet soda? Do the people who drink diet soda also have other unhealthy behaviors, like not exercising?) that we can’t use this data to prove that artificial sweeteners increase appetite or cause weight gain.
2: Do artificial sweeteners trigger the body’s hormonal response, similar to ingesting sugar? The short answer is that there appears to be no clinically significant difference in glycemic response (how blood glucose levels rise after a meal), gut hormones (such as GLP-1), or insulin. I say clinically significant rather than statistically significant because there was a paper that showed an increase in blood glucose (BG) levels with sucralose, but it was a very small amount – one that would not be noticed by your doctor because it is not big enough to be important. There continues to be research done on the topic, especially with all of the new artificial sweeteners on the market today. (http://ajpgi.physiology.org/content/296/4/G735 – http://www.ncbi.nlm.nih.gov/pubmed/25271009)
3: Do artificial sweeteners cause you to crave other sugary food? Since you asked your question a few days ago, I’ve been researching this question. There have been more studies done using functional MRI machines to watch which part of the brain responds to either sugar (sucrose) or certain artificial sweeteners. The study I found (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3465626/) did find a difference in how the brain responds in people who are “regular” diet soda drinkers compared to non-diet soda drinkers (Note: there were also MAJOR limitations of the study – their definition of “regular” drinkers was >1 diet soda per week, but the intake varied widely. They also did not mention the subjects’ consumption of regular soda. They also used saccharin as the sweetener tested, even though diet soda is primarily sweetened with aspartame.)
My non-scientific answer, based on experience with patients, friends, and family is that our tastes are affected by what we eat most often. So, if you continually expose yourself to high-intensity sweeteners (whether real sugar, corn syrup, or artificial), your taste buds will be less sensitive to the sweetness of real food, like fruits and veggies, and you will probably crave sweets more often.
Sweetness is a flavor that we crave naturally (thus why breastmilk is sweet), so sweet cravings will never go away entirely. The key is to moderate this craving so that in our modern society (where high sugar contents abound) we don’t go overboard.
Hope this (very long) response answered your question. Please let me know if you have other questions! And thanks for reading. 🙂
Only just come across this article and, disappointedly find it misses the key point: neotame and advantame can be used without declaration.
Sweeteners always have to be declared, but if the function is initially said to be flavour enhancer then a retained function is more difficult to show & a classification of processing aid can be applied – and hence, no declaration of use.
I am well versed in the physiological chemistry of phenylalanine, and aware that the precise mechanism by which high levels (an oversimplification here) lead to mental retardation have not been fully elucidated. Without that knowledge we cannot be certain of ‘safety’ using phenylalanine as an isolate and in foods with no, or little protein. None of the ‘tests’ carried out so far properly address the complexities.
Therefore, consumers should always be told when either neotame or advantame has been used (aspartame is always declared) and be free to exercise a choice to avoid it.
Pam, I so appreciate your comments! Thank you for opening up this discussion. I hear a number of different concerns, so let me try to address each of them.
What I hoped to provide with this specific blog post are the basic facts about artificial sweeteners based on the most credible evidence available at this time. In my practice, I encounter many clients who have similar concerns as you with food safety and quality. As a Registered Dietitian I work to guide my clients towards the best diet for that individual, based on the most quality, up-to-date research that I have access to. I have tried to apply the same principle to this article.
I recognize there is bias inherent in all research, and I too am still learning. I truly appreciate you sharing your point of view, and would gladly look into your reasons for coming to the conclusions you have. If you have other evidence I haven’t included, please send it my way! I would love to read it so I can continue to inform my opinion. I want to continue to learn as I recognize that new evidence comes out all the time.
Declaration/Labeling (specifically of neotame/advantame): I think you might have to get me up to speed on this. From what I could find, both of these have the same labeling requirements as other artificial sweeteners. They are not required to be labeled specifically for avoidance in Phenylketonuria (see description in the point below) because the amount used to sweeten foods is such an incredibly small amount that it doesn’t affect blood levels of phenylalanine. (If a Diet Coke was sweetened with neotame, the phenylalanine content would be 1.4mg; if with advantame it would be about 0.5mg – compared to 1 egg which has 630mg phenylalanine).
Is this what you were referring to when you say that aspartame is always declared? Or are you saying these ingredients are not included on the food label? If so, could you share your references for this? I unfortunately couldn’t find good sources. (Here are my sources: http://www.fda.gov/Food/IngredientsPackagingLabeling/FoodAdditivesIngredients/ucm397725.htm and http://www.neotame.com/faq.asp)
Your concern about phenylalanine: I didn’t go into great deal about this amino acid, because it is largely not applicable to the general population. Let’s back up to talk about why this naturally-occurring building block of proteins is even being discussed as something harmful.
There is a unique, rare genetic disorder called Phenylketonuria (PKU) in which a person is unable to produce the enzyme phenylalanine hydroxylase which is responsible for converting phenylalanine into other metabolites. Phenylalanine is one of the 9 essential amino acids (AA), meaning that the body cannot produce this amino acid, and so it is essential that a person obtains phenylalanine from the diet. However, if a person is unable to metabolize this specific AA, the more phenylalanine a person eats, the more it builds up in the blood. (https://ghr.nlm.nih.gov/condition/phenylketonuria#)
If a person with PKU ingests too much phenylalanine, levels build up in the blood. This causes a variety of problems including mental retardation. You are correct that the exact mechanism of why this happens is unknown, but the known treatment to prevent ill effects of PKU is to follow a very strict, low-protein diet to avoid over-consumption of phenylalanine (http://www.mayoclinic.org/diseases-conditions/phenylketonuria/basics/treatment/con-20026275)
Living life with PKU means constantly balancing obtaining enough phenylalanine to live without having too much that it would cause deleterious effects.
The diet for PKU not only warns against artificial sweeteners like aspartame that contain phenylalanine, but also essentially prohibits milk, cheese, eggs, all kinds of meat, nuts, and beans – because they all contain phenylalanine (and have MUCH higher quantities than what you would find in a diet soda that is sweetened with aspartame).
To put this in perspective, 1 g aspartame = ~500mg phenylalanine (https://engineering.purdue.edu/brl/PKU/PheEst0.pdf).
A Diet Coke contains 125mg aspartame, so about 70 mg phenylalanine (http://static.diabetesselfmanagement.com/pdfs/DSM0310_012.pdf).
1 egg, by comparison, contains about 620 mg phenylalanine (http://apjcn.nhri.org.tw/server/info/books-phds/books/foodfacts/html/data/data2e.html#chart).
I recognize an argument against artificial sweeteners is that phenylalanine is consumed individually instead of in a “natural” balance with other AAs. However, most people don’t only consume Diet Coke all day long (if they do, I have something to say about that as well!). When a moderate amount of diet soda is consumed in combination with eating a normal protein-containing diet, the balance of AAs normalizes.
Here are two studies (albeit small, and I don’t have the entire paper) that show the effect of plasma phenylalanine levels in individuals with and without PKU:
https://www.ncbi.nlm.nih.gov/pubmed/2890728
https://www.ncbi.nlm.nih.gov/pubmed/1617863
Essentially there was not a clinically significant rise in phenylalanine levels in either group (although I would argue against giving diet soda to someone with PKU – why risk it?).
Outside of PKU, I was unable to find any reason that a person needs to watch the intake of phenylalanine. I did see a Mayo Clinic article written by an RDN that mentions needing to avoid aspartame if you struggle with anxiety, but I couldn’t find the source she cited for that and a quick Pub Med search didn’t turn up anything about this. (http://www.mayoclinic.org/healthy-lifestyle/nutrition-and-healthy-eating/expert-answers/phenylalanine/faq-20058361)
Do you know any of the research behind this? I’d be interested to learn more about this possible link.
I know this was a really long response; I tend to get a little wordy. Does this address your concern about levels of phenylalanine?
Also, I’m not sure if you saw the sister post to this article: https://peasandhoppiness.com/nutrition/2015/11/26/the-great-battle-on-artificial-sweeteners . In that, I speak more practically about my general recommendations about how people should consume artificial sweeteners in their everyday life, not as much related to the basic safety. My general view of artificial sweeteners is similar to my view on bacon: it’s not exactly good for you, but a little bit won’t hurt you.
Thank you again for your comment. I really appreciate your perspective and welcome further discussion, either here or in person!